I’ve decided to do a series covering the various methods we can use to determine the age of the earth. This is a key issue since most evolutionists use this as a bat to beat creationists with. In fact, millions or billions of years is one of their primary arguments against creation. It also is one of the primary reasons young people leave the church. This article will serve as a primer of sorts, and will use the example of C-14. We will get into the specifics of each method as we go on throughout this series.
What makes something acceptable as a dating method? And no, having good pick up lines does not qualify. What we are talking about here are geochemical clocks. These are chemical, geological and cosmological rates which we can look at to determine how old the earth and/or universe is. Not everything works well as a geochemical clock, however. Some processes have issues with them that make them a poor fit as a method to count backward to earth’s origin.
For a process to make a good geochemical clock, it needs to have a couple of characteristics. It needs to operate at a known rate. Obviously, if the rate of a process varies periodically, then it would be impossible to use it to extrapolate backward. Further, the initial condition of the earth must be known. It is impossible to do a calculation of the earth’s age if there is an unknown on both sides of the equation. That’s a basic mathematical principle. The process must also be irreversible. This kind of goes along with the idea of a constant rate. If you have two elements, as you do in radiometric dating, for example, it would be impossible to calculate how long they had been in a rock or fossil if they were constantly turning into each other. It has to be a one-way street. The last essential condition is that the end result is known. That one should be a no-brainer, but it has to be stated so that everyone is on the same page. If a dating method fails on any of these points, it is not adequate to explain the age of the earth.
Before getting into C-14 dating, however, I want to illustrate a point. Only the youngest dating methods matter. The oldest ones must be wrong, by definition I’m going to borrow an example from Dr. Kent Hovind, who, while I don’t agree with him on a lot, had excellent word pictures. Suppose you find a treasure chest with a lot of old coins in it and you want to know when it was buried. How do you find that out? Well, you look at the coins in the box. Do you look at the oldest coin or the newest one? The newest one of course. Looking at the oldest coins would be silly. They wouldn’t give you an accurate representation of when the box was buried because it had to have been buried after the newest coin was produced. It is the same with the earth. It had to have come into being at the same time as, or after the youngest clock. Keep that in mind.
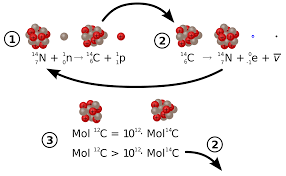
With those four rules in place, let us see how one of evolution’s iconic dating methods, Carbon-14 dating holds up to the scrutiny. The mechanism of carbon-14 dating is pretty simple. Carbon-14 is ingested by an organism while it is alive. However, Carbon-14 is unstable so it does not stay in the Carbon-14 state for long. Instead, it decays into the more stable Carbon-12. It does this at a known rate, called a half-life. The half-life of Carbon-14 is approximately 5700 years. There is an equation used for dating items using Carbon-14. It is as follows:
t = [ ln (Nf/No) / (-0.693) ] x t1/2 t
t=time
Nf =percent carbon on fossil
No =percent in the living organism
t1/2 =half-life
Thus a fossil with twenty percent of the original amount of Carbon-14 would have an equation as follows:
t = [ ln (0.20) / (-0.693) ] x 5,700 years
Therefore t= 13237.8 years would be the approximate age.
However, the above math has significant assumptions driving it. We did not start with a known amount of C-14. We started with a known fraction of the original. How do we know how much was in the original to derive our twenty percent? We have no clue how much C-14 was in the original substance. C-14 dating fails at the first hurdle. The initial condition is not known.
However, let us look further. Does C-14 decay happen at a known rate? Yes in the present. In the past, however, is another matter. In a catastrophic past, like even evolutionists are beginning to admit has plagued earth, the rate of radioactive decay could have been drastically sped up. Dr. Andrew Snelling has actually proposed this idea to coincide with the Flood. C-14 decay fails the constant rate test as well.
These are massive black marks against this particular form of radiometric dating. A further mark against it comes when you consider that some of the product and parent elements could potentially have leeched out of the rock in question. Since C-14 dating is dependent on measuring the end product and knowing the beginning product, it must be assumed that there was no loss of either beginning or end product. However, rocks are not closed systems. Water rushes across and through them regularly. This water could easily remove the parent or daughter elements, or even lodge some new daughter or parent element in the rock.
Carbon-14 dating is not used for dating anything older than about sixty thousand years as even evolutionists acknowledge there should be no C-14 in items older than that. However, as a creationist, I am going to make a prediction. I predict if they were to test the “oldest” marine fossils or dinosaur fossils, they would show C-14 levels and would thus date much younger than the millions of years assigned to them. The issues with C-14 dating do not end with the iconic dating method. They will crop up again and again as we discuss the various geochemical clocks. Buckle in, this could be a bumpy ride.